What did scientists discover?
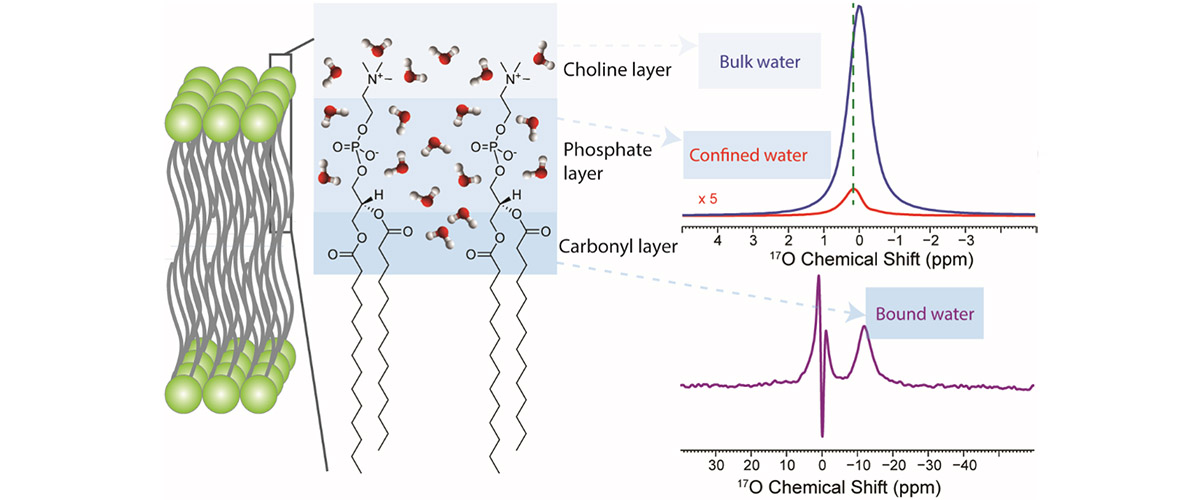
A new nuclear magnetic resonance (NMR) technique at 35.2T can identify 17O NMR signals of chemically and dynamically distinct water molecules. These signals are typically obscured by the large signals of bulk water.
Why is this important?
It is vital to understand the interactions of water molecules with proteins and membranes and their impact upon molecular structure and dynamics, as well as biological activity.
Water is ubiquitous and essential for the existence of all known life forms. Recently, it has been recognized that the mobility of water molecules in constrained biological systems is restricted and highly related to important biological functions. However, direct observation of these water molecules is challenging due to their extremely low populations compared to that of bulk water molecules. This new technique efficiently suppresses the large 17O NMR signals arising from bulk water molecules, thereby allowing for the detection of individual water molecules in a lipid bilayer environment, providing new opportunities to study biologically relevant water molecules in biological systems.
Who did the research?
Rongfu Zhang1,2, Timothy A. Cross1,2, Xinhua Peng3, Riqiang Fu1
1National MagLab; 2Florida State University; 3University of Science and Technology of China
Why did they need the MagLab?
High field magnets significantly improve both sensitivity and resolution of 17O NMR spectra of bound water molecules. The MagLab's 35.2T Series Connected Hybrid system, the highest-field NMR spectrometer in the world, is essential for this research.
Details for scientists
- View or download the expert-level Science Highlight, Finding Water Molecules with Important Biological Activity
- Read the full-length publication, Surprising Rigidity of Functionally Important Water Molecules Buried in the Lipid Headgroup Region, in Journal of the American Chemical Society
Funding
This research was funded by the following grants: G.S. Boebinger (NSF DMR-1644779); T.A. Cross (NIH GM122698)
For more information, contact Robert Schurko.