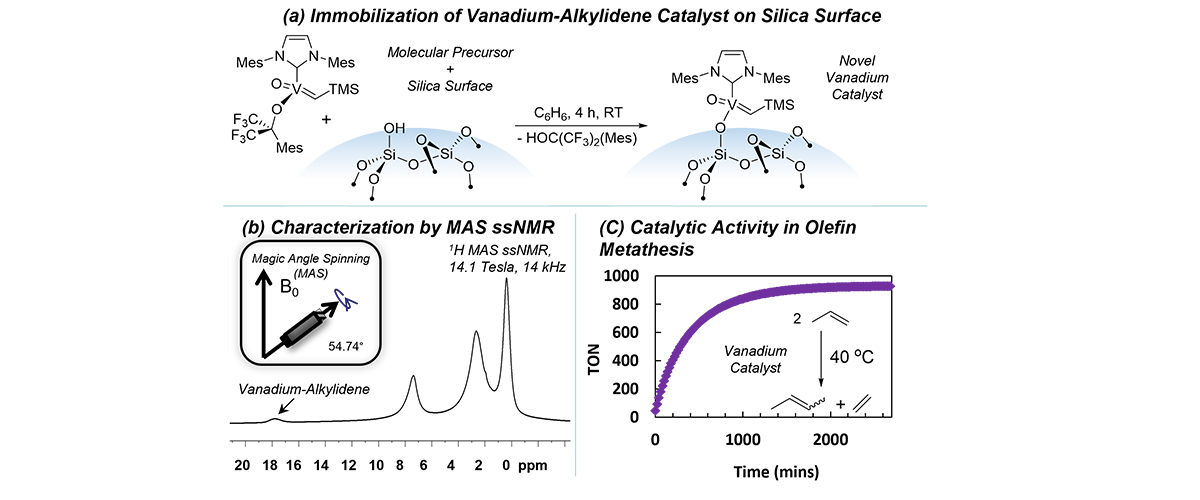
What is the finding
Researchers developed a new chemical catalyst based on vanadium, an earth-abundant, inexpensive metal, that breaks and forms carbon-carbon double bonds through a reaction called olefin metathesis. By attaching the catalyst to the surface of silica, the team dramatically improved its performance compared with the original molecular form. Advanced nuclear magnetic resonance measurements confirmed the structure of the active catalyst on the surface for the first time.
Why is this important?
Olefin metathesis is widely used to produce fuels, plastics, and other valuable chemicals. Effective catalysts typically rely on rare and expensive metals like molybdenum or tungsten. Developing catalysts based on abundant elements like vanadium could make these processes more sustainable and potentially less expensive. Many industrial catalysts operate on solid surfaces, but their atomic-scale structures are difficult to determine. By revealing how a vanadium catalyst functions when immobilized on silica, this research provides insight that could help scientists design more efficient catalysts for industrial processes.
Who did the research?
Rahul Koottanil Haridasan1, Shirley Hernandez2, Selena Kuenzig1, Gabriela Tejada2, James H. P. Collins3, Konstantin V. Bukhryakov2, Keith Searles1
1University of Florida; 2Florida International University; 3National High Magnetic Field Laboratory
Why did they need the MagLab?
Determining the structure of catalysts on solid surfaces requires high-field solid-state nuclear magnetic resonance spectroscopy, which can probe the atomic-scale environment of materials that cannot be studied in solution. The Advanced Magnetic Resonance Imaging and Spectroscopy Facility (AMRIS), affiliated with the MagLab, provides the high-field magnets and specialized instrumentation needed to perform these measurements.
Details for scientists
- View or download the expert-level Science Highlight, Olefin Metathesis Catalyzed by a Novel Surface Vanadium-Alkylidene
- Read the full-length publication, Silica-Supported Vanadium-oxo-alkylidene for Self-Metathesis of Propene, in Journal of American Chemical Society
Funding
This research was funded by the following grants: NHMFL (NSF DMR-2128556, NIH S10 OD028753); K. V. Bukhryakov (NIH R35GM150902)
For more information, contact Joanna Long.



![Metabolism pathways of [2H7]glucose (green stars), with red dots marking the presence of 2H, and larger dots indicating two 2H atoms. Note that HDO (gold stars) can be produced at multiple steps in the glycolytic pathway.](/media/mnyndfnd/august2020_amris_cancer_metabolism.jpg)