First some background
Some physicists explore the behavior and properties of specific molecules that have the potential to be used in future quantum computers, memory storage and other applications. In particular, they see promise in a property all molecules have called “spin,” which relates to its electrons. While electrons can, of course, move from one atom to another to conduct electricity in some materials, these negatively charged particles also are tiny magnets, oriented in a certain direction within their atomic framework. This is what physicists call “spin,” and these spins can have specific, discrete values (spin = 1 or spin = ½, for example) that influence a molecule’s magnetic properties. The nascent field of spintronics involves leveraging the spins of electrons, rather than current, to perform computer logic.
One enticing playground for physicists is the land of “low-dimensional materials,” which are very different than the 3D world we experience. These include 2-dimensional materials like 1-atom thick layers of graphene (a version of carbon), and even 1-dimensional materials made up of a string of molecules.
What did scientists discover?
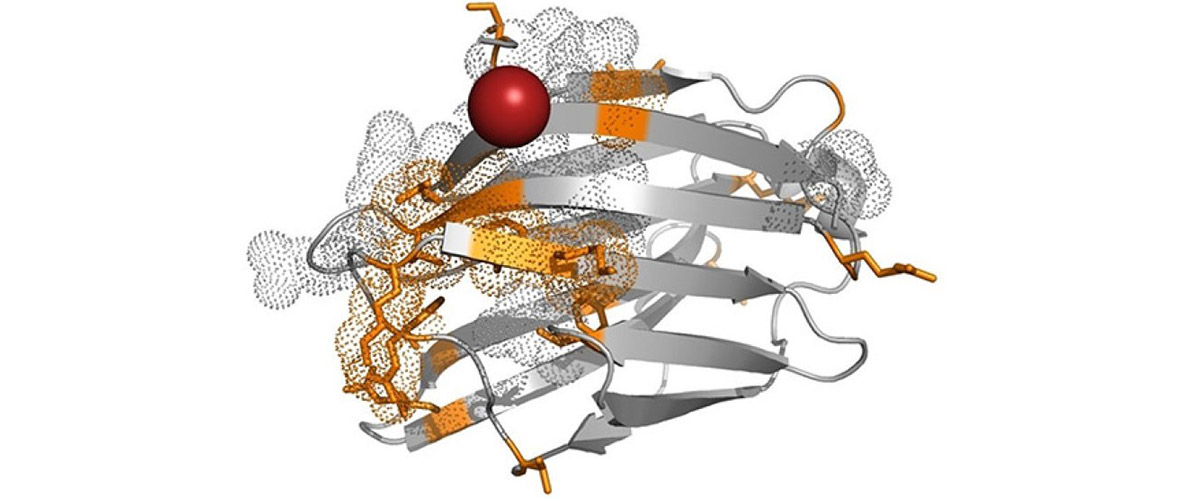
In this study, physicists experimented with a one-dimensional system made up of a chain of copper-based molecules with the chemical formula [Cu(pyrimidine)H2O4] SiF6 H2O. These molecules are strung together to create a 1-dimensional system resembling a kinked chain (see image above). Each of the four copper atoms has a differently oriented spin. When several of these 4-molecule units are attached together, the resulting chain turns in a corkscrew-like pattern.
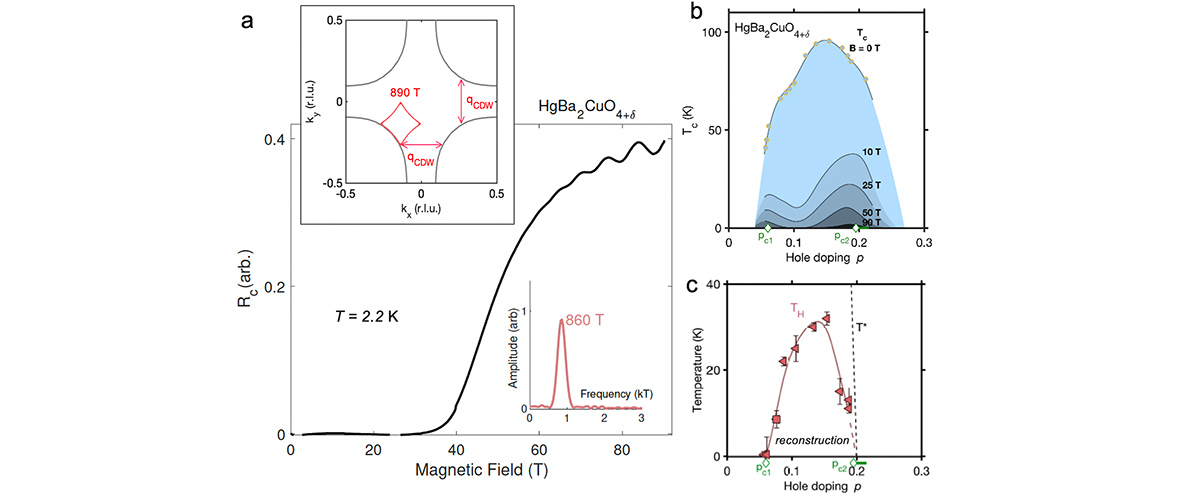
When scientists put this material in a high magnetic field, they discovered that it had unique magnetic properties that were not, based on its spin, predicted by theory.
Specifically, the material has a spin of ½, typical of copper. Such materials typically don’t exhibit a so-called “spin gap,” a feature that endows the system with magnetic properties. However, when the scientists put this material in a high magnetic field, it did form a gap, a behavior they attributed to its screw-like shape.
Why is this important?
This research provides a better understanding of the magnetism in this and other low-dimensional materials.
Who did the research?
J. Liu1, S. Kittaka2, R. D. Johnson1, T. Lancaster3, J. Singleton4, T. Sakakibara2, Y. Kohama2, J. van Tol5, A. Ardavan1, B. H. Williams1, S. J. Blundell1, Z. E. Manson6, J. L. Manson6, and P. A. Goddard7.
1Univ. of Oxford, UK; 2Univ. of Tokyo, Japan; 3Durham Univ. UK; 4National MagLab, Los Alamos; 5National MagLab, Tallahassee; 6Eastern Washington Univ.; 7Univ. of Warwick, UK.
Why did they need the MagLab?
This research required high magnetic fields and frequencies to study the spin excitations as a function of the magnetic field, capabilities uniquely available at the National MagLab’s Electron Magnetic Resonance
Details for scientists
- View or download the expert-level Science Highlight, Anomalous Spin Excitations in a Chiral Staggered Chain
- Read the full-length publication, Unconventional Field-Induced Spin Gap in an S = 1=2 Chiral Staggered Chain, in Phys. Rev. Lett.
Funding
This research was funded by the following grants: Funding Grants: G.S. Boebinger (NSF DMR-1157490, NSF-DMR1644779); P.A. Goddard (EPSRC, ERC-681260); J.L.Manson (DMR-1703003); R.D.Johnson (Royal Society)
For more information, contact Hans Van Tol.



![Scientists studied the copper-based compound [Cu(pyrimidine)H2O4] SiF6 H2O. Repeating units of four copper ions (see above) create corkscrew-like chains with intriguing magnetic properties.](/media/ayslgnyz/june2019_emr_chiral_staggered_chain.jpg)