At the end of the day, science is all about sharing: sharing ideas, discoveries, techniques, successes and failures. After all, exciting insights or results aren't worth the bits used to record them if they never make it off a researcher’s hard drive.
Paul Canfield, a physicist at Ames Laboratory and Iowa State University for more than two decades, knows that as well as anyone.
Canfield researches the design, discovery, growth and characterization of novel electronic and magnetic materials, and has made key contributions to our understanding of superconductivity, heavy fermions, quantum criticality and other phenomena. Over the course of his career, he has crossed paths with thousands of fellow scientists by co-authoring papers, mentoring students, and traveling the world to train others on his techniques. Science must be social, he said: That's how ideas spread, disciplines cross-fertilize and transformative science happens.
"My feeling is, at a certain point, you're much better served working with people who a) might be able to bring different or new takes and ideas, or b) are experts at what they do," Canfield said. "To understand the many aspects and facets of a new material, you need to appreciate it holistically."
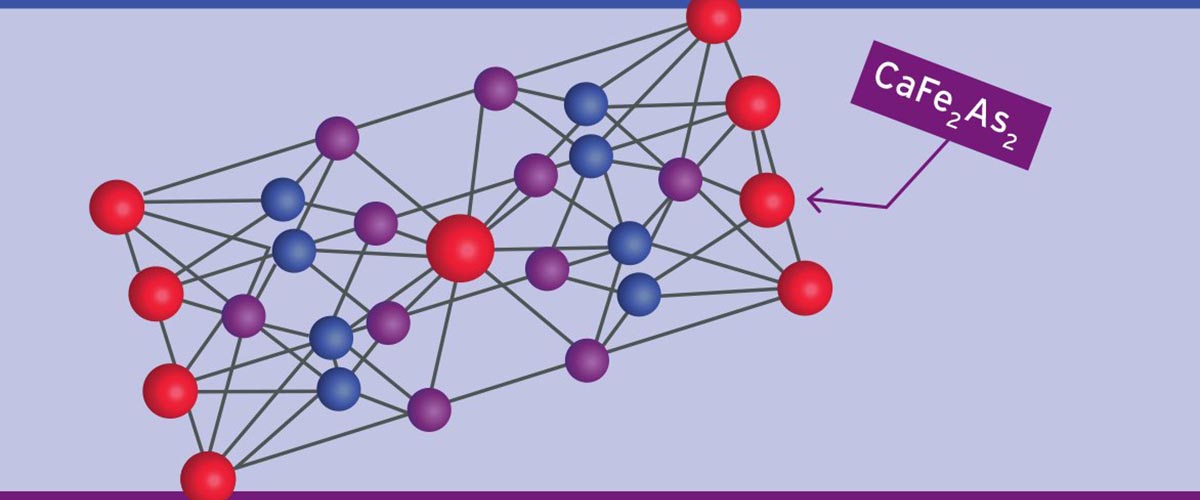
This high-impact scientist sits at the center of a complex web of physicists and chemists spanning generations and continents, each entangled in his or her own professional network. The many overlapping relationships are far too complex to represent in any diagram. Nevertheless, we have taken a modest stab at doing just that on this page, using one of Canfield's well-studied crystals, calcium iron arsenide (CaFe2As2), as the lattice structure for this "web of science." Though far from exhaustive, it at least gives a sense of the very collaborative nature of science.
Text by Kristen Coyne. Illustrations by Caroline McNiel